Acute Dental Pain
Acute pain is typically sudden and brought about by some type of injury or event such as muscle strain or a broken bone. It is characteristically sharp in its quality and will heal over time as the injury heals. Another hallmark of acute pain is its duration. Many doctors and physicians can determine the difference between acute and chronic pain by the amount of time a person has had a specific pain. Acute pain can last as long as six months but usually will subside sooner.Types of acute pain can include:
- Traumatic pain: broken bones, burns, cuts, etc.
- Surgical pain: pain stemming from recent surgery, typically felt during the recovery process
- Muscle strain: Injuries from sporting events or physical activity
Treatments for acute pain can range anywhere from over-the-counter drugs such as ibuprofen and acetaminophen to physical therapy or exercise. For more serious acute pains, such as traumatic pains from broken bones or surgery may require more intensive treatments or therapies.
The International Classification of Orofacial Pain sub categorises acute trigeminal pain as; Orofacial pain attributed to disorders of dentoalveolar and anatomically related structures with subcategories;
- Dental pain Pulpal pain ((dentine sensitivity, post trauma or treatment)
- Periodontal pain
- Gingival pain
- Oral mucosal pain
- Salivary gland pain
- Jaw bone pain
Acute trigeminal pain is a common presentation in the dental surgery. This article discusses the mechanisms underlying the pain experience, diagnosis and subsequent management of the acute trigeminal pain, encompassing pre, peri and postoperative analgesia.
Please see video lecture below and PDF lectures for further information.
Diagnosis Menu
Any patient attending the dentist will be experiencing some degree of anxiety and stress, these emotions will lower the patients’ pain tolerance and further compound their management. Managing operative pain under local anaesthesia requires expertise, empathy and patience. Anyone in this field recognises that pain is complex and particularly in the dental environment, where fear, phobia and poor expectations compound the patient’s pain experience, dentists require an armamentarium of psychological, communication, medical and technical skills rarely required in other branches of clinical medicine. Acute pain management is integral to the provision of optimal dental care and supporting the well-being of patients. Oral analgesics are commonly prescribed for a few days following oral surgery or other procedures, after which patients are typically pain-free or can switch to over-the-counter medications (i.e., either lower doses of the same analgesics or different OTC drugs).
Acute trigeminal pain is a distressing, common encompassing pain from the orofacial region and head (1). A cross-sectional population study in Cheshire, England, reported that orofacial pain (OFP) affected a quarter of the population, of whom only a half sought help (2). The prevalence was higher in women and younger adults (18-25 years) with 17% of the population having time off work or couldn’t carry out normal activities due to the pain (2). The impact of pain on the economy is demonstrated by a cross-sectional survey in eight countries in Europe which estimated the total annual cost of headache among adults, aged 18 to 65 years, as €173 billion (3).
The dental profession, since its infancy, has been a pioneer in the fields of anaesthesia and pain control. This stems from the need for these modalities in order to render pain free dental care in an anatomic region that is highly innervated by the second and third divisions of the trigeminal nerve. Pain has a dramatic physiologic impact that can adversely affect the health and well being of dental patients. Pain can have a profound effect on the cardiovascular, pulmonary, endocrine and gastrointestinal systems. Furthermore, if acute pain is not treated adequately, there is a risk that it may become chronic in nature. Therefore, adequate pain control is a medical and dental necessity and not merely an issue of patient comfort.
It is now understood that early control of acute pain can shape its subsequent progression, by preventing nociceptive input and hence, preventing persistent pain (4). Good pain management can help prevent the negative physiological (tachycardia, hypertension, myocardial ischemia, decrease in alveolar ventilation, and poor wound healing) and psychological (anxiety, sleeplessness, phobia) outcomes (5).
Management of acute dental pain includes management of patients undergoing surgery (peri and postoperative) and those presenting with pain as a result of underlying pathology (e.g. pulpitis, ulcer). Patients with trigeminal pain often present to dental practitioners. Successful management of acute trigeminal pain is dependent on obtaining a correct diagnosis of the source of pain(6). This is achieved through comprehensive history taking, examination and appropriate special tests (Table 1). Initial history taking should include determining the site, onset, character (type of pain), severity (verbal/ numerical, Table 3) and any exacerbating/ relieving factors. A thorough assessment can be completed taking into account associated signs and symptoms, radiation, functionality, disability, psychological effects and time course. Management of acute pain (7) as a presentation symptom is discussed in later sections.
Management of acute trigeminal pain can be divided into three areas – intra operative, post operative and acute symptomatic pain (usually acute infection).
- Management of intra operative pain Local anaesthesia with or without adjunctive sedation (for anxiolysis) and general anaesthesia for prolonged or extensive surgical procedures. Management of anxiolysis, non medical (behavioural) and medical (sedation) is not covered in depth in this section.
- Management of post surgical pain
- Management of acute orofacial pain-patient presenting with dental pain as a symptom
Pain Management: Part 1: Part 1: Managing Acute and
Postoperative Dental Pain
The painful tooth: mechanisms, presentation and differential diagnosis of odontogenic pain
Acute Pain Management: Scientific Evidence
Mechanism, peripheral mediators, central modulation and the trigeminal anatomy of pain have been covered in other sections of the website
Pain assessment
The clinician is beholden to take a full and comprehensive history to build trust and understanding of the patient and their complaint. Acute pain will have onset in the last days weeks and generally has been present for less than 3 months. The pain may be associated with key inflammatory signs (tumor dolor calor rubor and loss of function) but if caused by a ‘cold bacteria’ may not have the traditional inflammatory signs (dry socket for example). Acute pain is inflammatory pain responding to to anti inflammatories (for example paracetamol and NSAIDs) and antibiotics if related to an infective cause.
There is no absolute measure of pain as it is a purely subjective experience. However, pain assessment is essential in diagnosing and monitoring patients’ response to treatment. Pain rating scales are often used in assessing pain intensity. They are quick and easy to use (Table 3) (7), whereas pain questionnaires can often assess the quality and character of the pain (e.g. McGill Pain Questionnaire (MPQ) (8)) as well as its intensity (8). The MPQ consists primarily of three major classes of word descriptors; sensory, affective and evaluative to specify the pain experience (7).
Intra operative pain management
Local anaesthesia (LA) is fundamental for pain control in outpatient oral surgery and dental procedures. Local anaesthetic is defined as a drug which reversibly prevents transmission of the nerve impulse in the region to which it is applied, without affecting consciousness (9). It is frequently used to control intra-operative and immediate postoperative pain. During acute tissue trauma mediated pain (e.g. tooth extraction), local anaesthetic blocks the pain signal transmission to the cerebral cortex, preventing pain perception and processing. Hence, local anaesthetic should always be used, even if the patient is under general anaesthesia as it prevents nervous system sensitisation and hence, reduces postoperative pain (10).
Local anaesthetic agents bind to sodium channels on axons, thus inhibiting the rapid passage of sodium ions and propagation of an action potential. There are 2 theories of the mechanisms of action of local anaesthetic both of which involve perturbation of the nerve cell’s sodium channels and hence, prevention of nerve depolorisation and firing. The membrane expansion theory suggests nonspecific swelling of the cell membrane by absorption of the LA whilst the newer specific binding theory describes binding of LA to a specific binding site of the sodium channel. Discovery of specific drug binding sites allows for the possibility of developing anaesthetics with greater sensitivity for specific sodium channels with reduced side effects (11).
Lidocaine is the most commonly administered local anaesthetic by dental practitioners although other available solutions (prilocaine, mepivacaine and articaine) offer advantages in certain situations (Table 4). In the severely medically-compromised patient, such as those with unstable angina, an adrenaline-free solution such as 3% prilocaine with felypressin should be used (12). In a meta-analysis, articaine 4% was found to be superior to lidocaine 2% in anaesthetising lower first molars (13).
Although there is limited scientific studies that local anaesthetics with higher concentrations (4%) result in higher incidence of paraesthesia (14), there remains a trend to avoid articaine 4% use in inferior alveolar nerve blocks. Local anaesthetic related lingual nerve injury is most likely to occur when multiple inferior alveolar nerve blocks are given, regardless of the local anaesthetic agent used (15). Importantly buccal infiltration Articaine will most likely avoid inferior dental blocks with Lidocaine for most of dentistry also reducing te likelihood of IAN LA related injuries.
Studies have suggested benefit in using bupivacaine (Marcaine), a long acting local anaesthetic, to limit postoperative pain following third molar surgery (16, 17) and endodontic treatment (18). Reports range from a reduction in pain at 8 hours (17) to 7 days (16) postoperatively. It is important to note that bupivacaine exhibits this property only when used as a nerve block and not when used as an infiltration. Levobupivacaine, is a newer anaesthetic solution with an improved safety profile than, and equivalent efficacy to, bupivacaine (the latter has rare reports of causing severe central nervous system (CNS) and cardiovascular adverse effects)(19). A double blind study showed similar anaesthetic efficacy between bupivacaine (0.5%) and levobupivacaine (0.5%) in inferior alveolar nerve blocks, suggesting that levobupivacaine is a useful alternative to bupivacaine due to its lower toxicity(20).
Anxiolysis
Cognitive and psychological factors are reported to play a significant role in the severity of reported postsurgical pain (21). If patient anxiety prevents the ability to comply with dental procedures under local anaesthesia, then anxiolysis can be provided using oral, inhalational or IV sedation techniques. General anaesthesia should be considered if local anaesthesia is contraindicated.
Pre-operative analgesia
Failure of inferior alveolar nerve blocks to anaesthetise teeth with symptomatic irreversible pulpitis (“hot pulps”) is partly due to inflammatory prostaglandin production inducing peripheral nociceptor sensitisation and central sensitisation (22). Studies have demonstrated that the use of ibuprofen and paracetamol before the inferior alveolar nerve block does not statistically improve success rate (22, 23). This is likely due to the fact that sensitisation has already taken place, and NSAIDs cannot reduce the amount of prostaglandin already present but rather may limit further production.
Preventive/ pre-emptive analgesia is a treatment that is initiated before the surgical procedure to prevent nociception and, hence, sensitisation of the peripheral and central pathways (24, 25). Although there are many studies in general surgery reporting the benefits of pre-emptive analgesia in reducing the postoperative pain experience, there are limited studies in relation to dental surgery (see section on NSAIDs) with some studies reporting no benefit (26). Dahl et al. 2011 highlight the need for improved design of clinical studies in order to achieve more conclusive answers regarding the different preventive interventions (24). Figure 1 Diagram of where and how OTC analgesics work here.
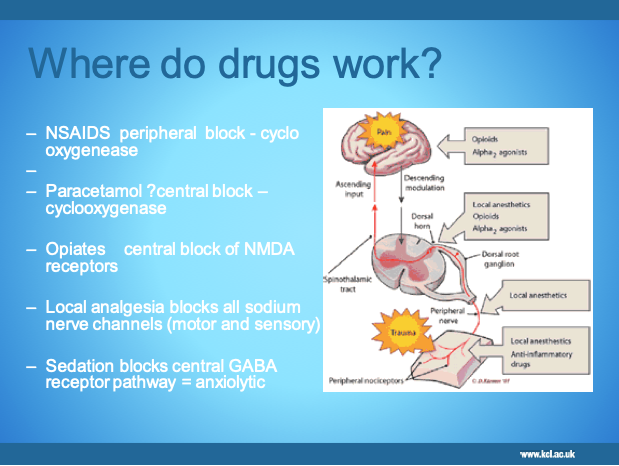
Post surgical pain management
Effective postoperative pain management is fundamental to quality dental care, and is likely to speed recovery. Conventional analgesics act by interrupting ascending nociceptive information or depressing interpretation of the information within the CNS. Oral analgesics are commonly prescribed for a few days following dental surgery, after which patients are typically pain-free or can switch to over-the-counter (OTC) medications (i.e., either lower doses of the same analgesics or different OTC drugs).
Analgesics are classified as opioids or non-opioids and may act at different sites depending on their mechanism of action. Non-steroidal anti-inflammatory drugs (NSAIDs) decrease pain resulting from inflammatory reactions (arachidonic acid cascade). Opioids may affect emotional aspects of pain and can modify transmission of pain information in the dorsal horn (descending modulation). Non-opioid analgesics (including paracetamol and NSAIDs) have been demonstrated to be superior analgesics in dental pain compared to opioids at conventional doses (25).
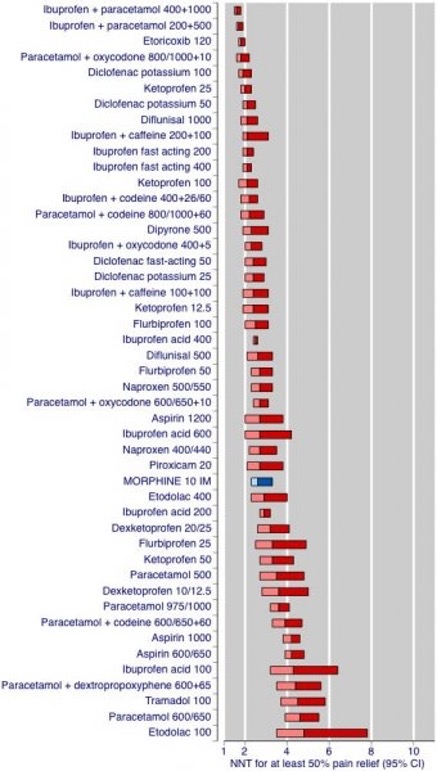
A meta-analysis of Cochrane reviews of randomised controlled trials (RCTs) testing the analgesic efficacy of individual oral analgesics in acute postoperative pain has helped facilitate indirect comparisons between oral analgesics (27). The results from this review and previous systematic reviews of randomised postsurgical analgesic trials helped to formulate the Oxford League Table of Analgesic Efficacy (28), which is used by healthcare professions worldwide (Figure 1 or table?). Analgesic efficacy is expressed as the number-needed-to-treat (NNT). This estimates the number of patients who need to receive the analgesic for one to achieve at least 50% relief of pain compared with placebo over a six-hour treatment period. The more effective the analgesic, the lower the NNT. The table shows that oral NSAIDs perform well, and that paracetamol in combination with an opioid is also effective.
NSAIDs.
NSAIDs are known for their analgesic, antipyretic, and anti-inflammatory properties. These therapeutic effects, as well their associated side effects, are mostly due to NSAID inhibition of the enzyme cyclo-oxygenase (COX) and hence prostaglandin (PG) production. Following trauma/surgery, arachidonic acid is released from phospholipid bilayers in perturbed cell membranes by the activated enzyme phospholipase A2. COX enzyme then catalyzes the formation of prostaglandins and thromboxane from arachidonic acid in the arachidonic pathway (see figure 1). Prostaglandins are involved in inflammation, nociception and fever modulation (prostaglandin E2 in the hypothalamus).
NSAIDs are the gold standard analgesics in dentistry as acute postoperative dental pain is inflammatory in nature, and hence are superior to opioids (29). Ibuprofen 200/400mg or diclofenac 25/50mg, three times daily, are commonly prescribed NSAIDs.
NSAIDS are broadly classified as non-selective cyclo-oxygenase (COX) -1, 2 enzyme inhibitors (ibuprofen, diclofenac, aspirin) or selective COX-2 enzyme inhibitors (celecoxib, rofecoxib). The non-selective group is further subdivided based on its derivative compounds.
NSAIDs are contraindicated for patients who have a history of gastrointestinal (GI) ulcer/erosions, anticoagulant therapy/ bleeding disorders, nephropathy, or intolerance/ allergy to such drugs (Table 4). If NSAIDs are contraindicated, paracetamol may be used as an alternative (see below).
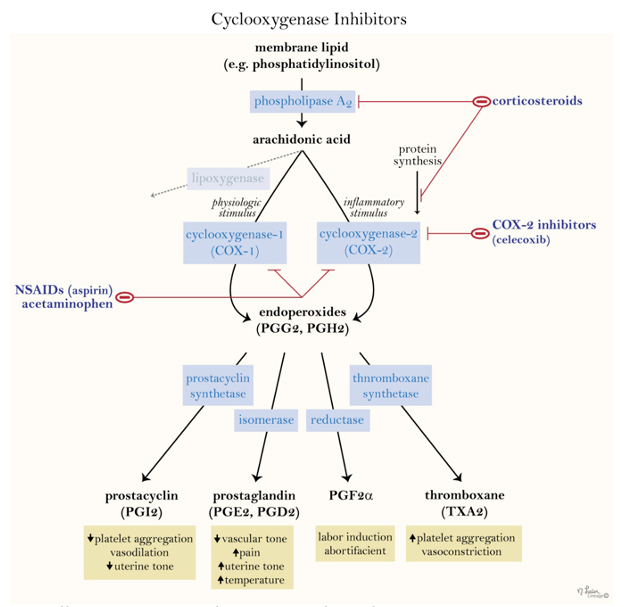
(https://step1.medbullets.com/derm-and-msk/112053/nonsteroidal-anti-inflammatory-drugs-nsaids)
Preoperative/ preventive use of NSAIDs has been demonstrated to decrease the intensity of postoperative pain and swelling (23, 30). During surgery the synthesis of prostaglandins at the surgical site will transmit nociceptive impulses and sensitise the nervous system to the pain. Administering NSAIDs before surgery will inhibit prostaglandin synthesis and reduce the postoperative pain experience once the local anaesthesia wears off. Optimum serum levels of NSAID should be established whilst the tissue remains anaesthetised for maximum benefit (29). Preoperative ibuprofen has been shown to be more effective at reducing acute postoperative pain than paracetamol or paracetamol with codeine, although the paracetamol was at suboptimal dose of 600mg (as opposed to 1g) (30).
Paracetamol
Paracetamol is one of the most popular and widely used drugs for first-line treatment of fever and pain (Bertolini et al., 2003). Paracetamol does not demonstrate significant anti-inflammatory properties, implying a mode of action that differs from that of NSAIDs (27) how ever it crosses the blood brain barrier.
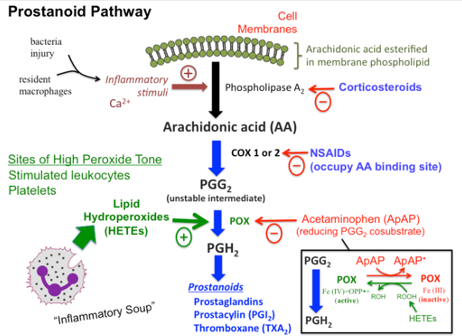
https://tmedweb.tulane.edu/pharmwiki/doku.php/acetaminophen)
Paracetamol is commonly prescribed as 1g, four times daily. Overdose of paracetamol can cause hepatotoxicity and death from liver failure, so clear instructions on dose and timing should be given to patients (31).
Although the efficacy of paracetamol is well established, its mode of action is still poorly understood. Paracetamol’s significant anti-pyrexial activity, suggests that the drug acts centrally. The following 5 mechanisms have been suggested: inhibition of cyclo-oxygenase isoenzymes (COX1,2,3), interaction with the serotoninergic inhibitory descending pathway, the endogenous opioid pathway (primarily descending), increase in cannabinoid/ vallinoid tone and involvement in the nitric oxide pathway (32). A recent study at King’s College London demonstrated, in the rat, that spinal pain receptors TRPA1 have a crucial role in the antinociceptive effects of paracetamol (33). Peripheral TRPA1 nociceptors did not demonstrate the same involvement (33).
Paracetamol is a safe drug, which is well tolerated, with minimal side effects. It is metabolised by the liver and is hepatotoxic. Hence caution is required in its use in chronic alcoholic patients and those with liver damage (Table 5). A Cochrane review, in 2007, of RCTs demonstrated that paracetamol is an effective drug to use for postoperative pain following oral surgery, with very few adverse effects (34).
Opioids
Opioids bind to specific opioid receptors in the central nervous system (CNS), causing reduced pain perception and reaction to pain and increased pain tolerance. In addition to these desirable analgesic effects, binding to receptors in the CNS may cause adverse events such as drowsiness and respiratory depression. Moreover, binding to receptors elsewhere in the body (primarily the GI tract) commonly causes nausea, vomiting, and constipation. In an effort to reduce the amount of opioid required for pain relief, and so reduce undesirable side effects, opioids are commonly combined with non-opioid analgesics. On this basis the opioid group of medication is not the first choice for management of mild to moderate acute orofacial pain (32). Since the recent opiate opioid crisis in the US these drugs are not prescribed for routine pain control. They are infact very weak in managing most post-operative pains. It should be noted that according to the league table opioids may be less effective for acute pain than medicines with other different mechanisms of action. In addition, there are significant side effects of opioids and opiates.
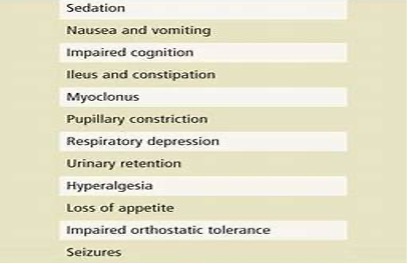
Selecting analgesics according to WHO analgesic ladder
An analgesic ladder, for differing pain severity, was introduced by the World Health Organisation in 1986 to assist analgesic prescribing by clinicians (Figure 2). Non-opioid analgesics (paracetamol and NSAIDs) form the basis of managing mild pain with introduction of opioid analgesia if the pain worsens (35). This principal of multi-modal analgesia highlights that the gold standard of pain management is by combinations of drugs, thereby maximising analgesic efficacy at lower doses and minimising side effects. It also advocates regular administration of analgesics (every 4 to 6 hours) rather than “on demand” (35).
The optimal pain management for toothache and post-surgical pain or related conditions is NSAIDs should be combined with paracetamol, when possible, as they provide greater analgesia than when used alone (32), reducing the effective dose and hence, possible side effects. This synergistic effect is attributed to different sites of action of the two analgesics (29). The initial prescription of paracetamol is often supplemented by oral nonsteroidal drugs. The latter is used pro rata (PRN- when necessary) as the pain decreases. Taking paracetamol with NSAID only when necessary can limit potential side effects of the NSAID.
Table 6 shows the authors’ suggested analgesic regime for acute trigeminal pain.
It is generally suggested that an optimal dose of 400-600mg oral Ibuprofen combined with 1000mg Paracetamol (or acetometaphen) see the table below (https://fpm.ac.uk/opioids-aware-clinical-use-opioids/opioids-and-acute-pain-management). *NNT (number needed to treat). The number of patients needed to be treated for one to benefit compared with a control. A treatment that works for everyone, and where no patient has a response with control, would have a NNT of 1. The higher the NNT, the less effective the treatment.
Management of acute odontogenic pain
See following links:
The painful tooth: mechanisms, presentation and differential diagnosis of odontogenic pain
Acute Pain Management: Scientific Evidence
Odontogenic pain refers to pain initiating from the teeth or their supporting structures, the mucosa, gingivae, maxilla, mandible or periodontal membrane.
‘A toothache, or a violent passion, is not necessarily diminished by our knowledge of its causes, its character, its importance or insignificance.’ T. S. Eliot
Aetiology of acute orofacial pain Orofacial pain can be associated with pathological conditions or disorders related to somatic and neurological structures. There are a wide range of causes of acute orofacial pain conditions, the most common being dental pain (toothache). Dental disease of the hard tissues (caries of enamel, dentine, and cementum), and soft tissues and supporting bone (gingivitis/periodontitis) are recognized as the most common diseases to afflict the general population (36). These conditions are largely diagnosed and treated by dental practitioners by history, dental clinical examination, and radiographs. By far the most common forms of oral pain are them acute form of pain that tend to last for short periods of time. These include toothache (dental pulpitis), gum pain (pericoronitis in 80% of the population) (37), periapical periodontitis (owing to apical infection or postendodontic therapy of high occlusal contact) (38). Dentine sensitivity affects 40% of the adult population; dry socket is postsurgical intense pain that affects 10% of patients after extraction of their teeth. Other orofacial acute pain conditions include trauma or infection of the orofacial tissues.
Toothache is caused by inflammation of the dental pulp (Figure 3) as a result of dental caries (tooth decay), the most common human infective disease worldwide, affecting 60-90% of school children worldwide. Interestingly periodontal disease (gum disease) the second most common infection is painless similar to other chronic mycobacteria infections for example Leprosy. Severe periodontal (gum) disease, which may result in tooth loss, is found in 5-20% of middle-aged adults; the rate (WHO Health statistics (39)).
- Dentofacial pain is a common presentation in general practice, and more than 50% of cases arise from dentally related pathology (40).
- In a carious tooth, pain that is site-specific, severe and spontaneous usually denotes extension of caries into the tooth pulp.
- Caries does not always appear as a cavity in the tooth, but may lie beneath intact enamel or on surfaces between teeth.
- Examination of dental pain should include firm percussion (eg, with a tongue depressor). Tenderness on percussion denotes progression of infection into theperiapical tissues.
Prevalence
The prevalence of dental pain and its characteristics were recorded using standard measures of pain (WHO criteria (39)). In a study of 1,052 individuals the prevalence of reported toothache in schoolchildren in the last six months was 33.6% (31.1-36.8, 95% CI). Multiple logistic and ordinal polytomous regression analysis showed a significant relationship between lower social class, later birth order, failure at school and attendance at the dentist only when in trouble with both the prevalence and severity of dental pain. The major predictor of the prevalence and severity of pain was pattern of dental attendance (p<0.001) (40).
Dental pulpitis (‘toothache’)
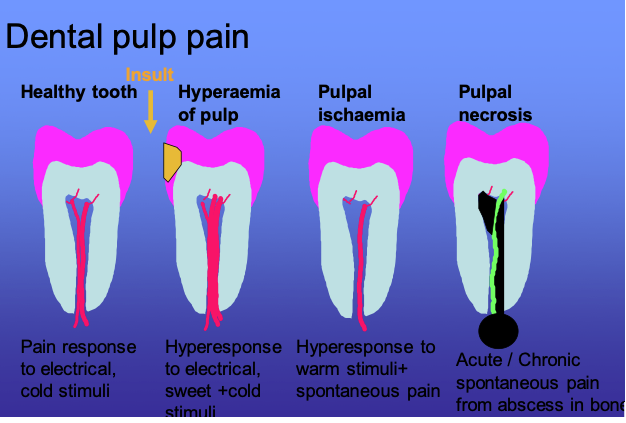
In health teeth are only perceive pain often due to dentine sensitivity on cold, sweet or physical stimulus. Dental pulpitis may be due to infection from dental caries close to the pulp, inflammation caused by chemical or thermal insult subsequent to dental treatment, and may be reversible or non-reversible. Intermittent sharp, shooting pains are also symptomatic of trigeminal neuralgia, so care must be taken not to mistakenly label toothache as neuralgia. Confirmation of the type of pulpitis is a clinical diagnosis (Fig 3).
If the insult persists the pulpitis will become irreversible with increased pulpal vascularity and resultant pressure, inducing ischaemia causing sensitivity to heat with prolonged pain. Once necrosis of the dental pulp has occurred, the infection spreads through the apex of the tooth into the surrounding bone and periodontal membrane, initiating periodontal inflammation and eventually a dental abscess causing spontaneous long lasting pain on biting on the tooth. Typically the pain associated with an abscess is described as spontaneous aching or throbbing and if associated with swelling in the jaw, trismus or lymphadenopathy it may be indicative of an acute spreading infection. Thus different stages of infection have different clinical presentations (Figure 3).
Figure 3 The stages and characteristics of dental pulpal pain.
Management Protection of the pulp to bacterial infection and chemical irritation by dietary and salivary content must be undertaken promptly to minimise persistence of acute pulpitis thus evolving into chronic irreversible pulpitis. This treatment will involve a filling or restoration which will resolve reversible pulpitis. Irreversible pulpitis may be managed in acute episodes using non steroidal anti inflammatory drugs (NSAIDs) during acute phases but the patient is better off arranging root canal treatment of the tooth to prevent further pain.
Exposed cementum or dentine There is tooth sensitivity from cold fluids and/or air, a reflection of a healthy pulp. With gingival recession, recent scaling, or tooth wear due to a high acid diet or gastric reflux, there may be generalised dentine sensitivity, management is summarised in Table 6.
Apical pain can be caused by infection spreading through the apical foramen of the tooth into the apical periodontal region causing inflammation (apical periodontitis) and ultimately a dental abscess if left untreated. This is treated by either root canal treatment of the tooth or extraction of the tooth with or without concomitant courses of antibiotics (Abbott 2000). Iatrogenic apical pain may result after dental treatment including premature contact if a restoration is left high in occlusion. This is characterized by an initial sharp pain which becomes duller after a period. The pain is due to a recent tooth restoration that is ‘high’ compared with the normal occlusion when biting together and will persist until the height is reduced. Apical pain pay also be induced postendodontic treatment, This is severe aching pain following endodontic treatment such as root canal therapy or apicectomy. While the majority of patients improve over time (weeks), a few will develop a chronic neuropathic pain state (see section on persistent post surgical trigeminal pain).
Dental management for dental abscess is either root canal with removal of the necrotic pulp or tooth extraction.
See New International Classification of Orofacial Pain: What Is in It For Endodontists?
Periapical inflammation can lead to a cellulitis of the face characterised by a rapid spread of bacteria and their breakdown products into the surrounding tissues causing extensive oedema and pain. If systemic signs of infection are present, for example, fever and malaise, as well as swelling and possibly trismus (limitation of mouth opening), this is a surgical emergency. Antibiotic treatment alone is not suitable or recommended. If pus is present, it needs to be drained, the cause eliminated, and host defences augmented with antibiotics. The microbial spectrum is mainly gram positive including anaerobes. Appropriate antibiotics would include a penicillin or a ‘first generation’ cephalosporin, combined with metronidazole in more severe cases.
Antibiotics may be prescribed prior to arranging necessary endodontic therapy or dental extraction. Metronidazole (200mg TDS PO 3-5 days) and or Penicillin V (250mgs QDS PO 3-5 days) are the antibiotics of choice. The analgesics of choice for dental inflammatory pain are ibuprofen (4-600mg Soluble QDS PO PRN) combined simultaneously with Paracetamol (Acetomephrin 500-1000mg QDS PO PRN) Table 5 provides more information about contrainidcations and side effects of these medications.
Pericoronitis Pain commonly arises from the supporting gingivae and mucosa when infection arises from an erupting tooth (teething or pericoronitis). This is the most common cause for the removal of third molar teeth (wisdom teeth). The pain may be constant or intermittent, but is often evoked when biting down with opposing maxillary teeth. This elicits pain in the inflamed mucosa and gingivae surrounding the partially erupted tooth. If recurrent, then pericoronitis is the main NICE indication for removal of wisdom teeth, however if the infection is acute and spreading then antibiotics must be prescribed. Chronic periodontitis with gradual bone loss, rarely causes pain and patients may be unaware of the disorder until tooth mobility is evident. There is quite often bleeding from the gums and sometimes an unpleasant taste. This is usually a generalised condition, however, deep pocketing with extreme bone loss can occur around isolated teeth. Food impaction in these areas can cause localised gingival pain. Poor contact between adjacent teeth and the presence of an occluding cusp forcing food into this gap can also cause a build-up of food debris and result in gingival inflammation.
Acute pericoronitis involves bacterial infection around an unerupted or partially erupted tooth and usually affects the lower third molar (wisdom tooth). The condition is often aggravated by the upper molar impacting on the swollen flap of soft tissue covering the unerupted tooth. There may be associated trismus due to the fact that the lower 8 site is close to tehmandibular insertion of the temporalis muscle, management is summarised in Table 6.
Alveolar osteitis After extraction, the most common complication is a ‘dry socket’ which is a condition whereby the clot formation within the socket fails at 3–5 days; healing fails, resulting in an empty socket which traps food and debris. The resultant pain is caused by necrotic foodstuff aggravating bony nerve endings, causing intense pain following extractions. Interestingly this condition is devoid of the usual acute inflamammatory markers (absence of lymphadenopathy, local inflammation and swelling). A dull throbbing pain develops two to four days after a mandibular tooth extraction. It rarely occurs in the maxilla. Smoking is a major predisposing factor as it reduces the blood supply. The tissue around the socket is very tender and white necrotic bone is exposed in the socket. Halitosis is very common. The incidence of this condition is between 1-9% associated with patients undergoing mandibular surgical tooth removal. Patients should be routinely warned of a possible incidence of 5%, management is summarised in Table 6.
Maxillary sinusitis ‘mimicking’ toothache Recurrent maxillary sinusitis may cause widespread pain in the maxillary teeth. The pain tends to be increased on lying down or bending over. There is often a feeling of `fullness’ on the affected side. The pain is usually unilateral, dull, throbbing and continuous. Quite often the patient feels unwell generally and feverish. It can mimic the maxillary sinusitis-like symptoms in temporomandibular disease (TMD) (see seperate section) or neuropathic pain. These dental conditions rarely present as chronic pain unless misdiagnosed. Inflammation of the maxillary sinuses is best treated using local and systemic decongestants and if persistent then antibiotics may be prescribed (42). Pain originating from the sinus arises mainly from pressure. Decongestants can help sinus drainage. Antibiotics probably have only a minor role in mild cases. Referral to an otorhinolaryngologist for endoscopic sinus surgery may be indicated in chronic cases (43).
Necrotizing ulcerative gingivitis (NUG) {formerly Acute necrotising ulcerative gingivitis} is a rapidly progressive infection of the gingival tissues that causes ulceration of the interdental gingival papillae. It can lead to extensive destruction. Usually young to middle-aged people with reduced resistance to infection are affected (diabetes, HIV infection, chemotherapy). Males are more likely to be affected than females, with stress, smoking and poor oral hygiene being predisposing factors. Halitosis, spontaneous gingival bleeding, and a `punched-out’ appearance of the interdental papillae are all important signs (44). The patients quite often complain of severe gingival tenderness with pain on eating and tooth brushing. The pain is dull, deep-seated and constant. The gums can bleed spontaneously and there is also an unpleasant taste in the mouth and obvious halitosis,
Non-odontogenic acute facial pain Non-odontogenic facial pain can be caused by inflammation due to tumour, infection, or trauma. Topographical classification is often applied to this complex region. Regions often presenting as orofacial pain complaints include the sinuses, salivary gland, ears, eyes, throat, mandibular, and maxillary bone pathology. Often in patients with chronic orofacial pain there will be multiple causes of discomfort including inflammatory odontogenic pain alongside a neuropathic, neurovascular or idiopathic cause these comditions are reviewed ins ections 3-8. Thus knowledge of the complexity of pain and local anatomy are crucial in order to diagnose and treat odontogenic causes of pain.whilst simultaneously excluding sinister causes of pain requiring urgent management or neuropathic pain in which patients will not nebefit from surgical or dental treatment.
Summary
Acute trigeminal pain is commonplace in the dental setting. Thorough history taking and special tests can accurately diagnose acute pain and allow subsequent management. Perioperative pain can often be well controlled using affective local anaesthesia, with or without anxiolytic, thereby negating the use of general anaesthesia. Preventive ibuprofen is beneficial if administered before the local anaesthetic wears off. Postoperative pain management is ideally controlled with combinations of non-opioid analgesics, namely ibuprofen/ diclofenac and paracetamol, for superior analgesia and limited side effects.
REMEMBER
Differential diagnosis of dental pain can be complex and if the patient des not respond to routine dental care, analgesics or antibiotics, then the dentists MUST reconsider a possible non odontogenic cause. This is highlighted in a recent paper here.
References
- Orofacial Pain From Basic Science to Clinical Management. Sessle, editor1999.
- Macfarlane TV, Blinkhorn AS, Davies RM, Kincey J, Worthington HV. Oro-facial pain in the community: prevalence and associated impact. Community Dent Oral Epidemiol. 2002 Feb;30(1):52-60.
- Linde M, Gustavsson A, Stovner LJ, Steiner TJ, Barre J, Katsarava Z, et al. The cost of headache disorders in Europe: the Eurolight project. Eur J Neurol. 2011 Dec 5.
- Carr DB, Goudas LC. Acute pain. The Lancet. 1999;353(9169):2051-8.
- Vadivelu N, Mitra S, Narayan D. Recent advances in postoperative pain management. Yale J Biol Med. [Review]. 2010 Mar;83(1):11-25.
- Hegarty AM, Zakrzewska JM. Differential diagnosis for orofacial pain, including sinusitis, TMD, trigeminal neuralgia. Dent Update. 2011 Jul-Aug;38(6):396-400, 2-3, 5-6 passim.
- Summers S. Evidence-based practice part 2: reliability and validity of selected acute pain instruments. J Perianesth Nurs. 2001 Feb;16(1):35-40.
- Melzack R. The McGill Pain Questionnaire: major properties and scoring methods. Pain. 1975 Sep;1(3):277-99.
- Edgcombe, Hocking. Local anaesthesia pharmacology. Update in Anaesthesia. 2005; www.anaesthesiologists.org.
- Gottschalk A, Smith DS. New concepts in acute pain therapy: preemptive analgesia. Am Fam Physician. 2001 May 15;63(10):1979-84.
- Meechan JG. Local anaesthesia. Oral Surgery. 2008;1(1):3-10.
- Meechan JG. How to overcome failed local anaesthesia. Br Dent J. 1999 Jan 9;186(1):15-20.
- Meechan J. Articaine and lignocaine. Evidence Based Dentistry. 2011.
- Meechan JG. Local anaesthesia: risks and controversies. Dent Update. 2009 Jun;36(5):278-80, 82-3.
- Renton T, Adey-Viscuso D, Meechan JG, Yilmaz Z. Trigeminal nerve injuries in relation to the local anaesthesia in mandibular injections. Br Dent J. 2010 Nov;209(9):E15.
- Nayyar MS, Yates C. Bupivacaine as pre-emptive analgesia in third molar surgery: Randomised controlled trial. Br J Oral Maxillofac Surg. 2006 Dec;44(6):501-3.
- Bouloux GF, Punnia-Moorthy A. Bupivacaine versus lidocaine for third molar surgery: a double-blind, randomized, crossover study. J Oral Maxillofac Surg. 1999 May;57(5):510-4; discussion 5.
- Moore PA, Dunsky JL. Bupivacaine anesthesia–a clinical trial for endodontic therapy. Oral Surg Oral Med Oral Pathol. 1983 Feb;55(2):176-9.
- Rood JP, Coulthard P, Snowdon AT, Gennery BA. Safety and efficacy of levobupivacaine for postoperative pain relief after the surgical removal of impacted third molars: a comparison with lignocaine and adrenaline. British Journal of Oral and Maxillofacial Surgery. 2002;40(6):491-6.
- Branco FP, Ranali J, Ambrosano GM, Volpato MC. A double-blind comparison of 0.5% bupivacaine with 1:200,000 epinephrine and 0.5% levobupivacaine with 1:200,000 epinephrine for the inferior alveolar nerve block. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. [Clinical Trial Randomized Controlled Trial]. 2006 Apr;101(4):442-7.
- Khan RS, Ahmed K, Blakeway E, Skapinakis P, Nihoyannopoulos L, Macleod K, et al. Catastrophizing: a predictive factor for postoperative pain. Am J Surg. 2011 Jan;201(1):122-31.
- Simpson M, Drum M, Nusstein J, Reader A, Beck M. Effect of combination of preoperative ibuprofen/acetaminophen on the success of the inferior alveolar nerve block in patients with symptomatic irreversible pulpitis. J Endod. 2011 May;37(5):593-7.
- Jackson DL, Moore PA, Hargreaves KM. Preoperative nonsteroidal anti-inflammatory medication for the prevention of postoperative dental pain. J Am Dent Assoc. 1989 Nov;119(5):641-7.
- Dahl JB, Kehlet H. Preventive analgesia. Curr Opin Anaesthesiol. [Review]. 2011 Jun;24(3):331-8.
- Dahl JB, Moiniche S. Pre-emptive analgesia. Br Med Bull. [Review]. 2004;71:13-27.
- Zacharias M, Hunter KM, Baker AB. Effectiveness of preoperative analgesics on postoperative dental pain: a study. Anesth Prog. 1996 Summer;43(3):92-6.
- Moore RA, Derry S, McQuay HJ, Wiffen PJ. Single dose oral analgesics for acute postoperative pain in adults. Cochrane Database Syst Rev. 2011;9:CD008659.
- Bandolier. Oxford League Table of Analgesia. 2007.
- Becker DE. Pain management: Part 1: Managing acute and postoperative dental pain. Anesth Prog. 2010 Summer;57(2):67-78; quiz 9-80.
- Dionne RA, Campbell RA, Cooper SA, Hall DL, Buckingham B. Suppression of postoperative pain by preoperative administration of ibuprofen in comparison to placebo, acetaminophen, and acetaminophen plus codeine. J Clin Pharmacol. 1983 Jan;23(1):37-43.
- Thomas MB, Moran N, Smart K, Crean S. Paracetamol overdose as a result of dental pain requiring medical treatment – two case reports. Br Dent J. 2007 Jul 14;203(1):25-8.
- Ong CK, Seymour RA, Lirk P, Merry AF. Combining paracetamol (acetaminophen) with nonsteroidal antiinflammatory drugs: a qualitative systematic review of analgesic efficacy for acute postoperative pain. Anesth Analg. 2010 Apr 1;110(4):1170-9.
- Andersson DA, Gentry C, Alenmyr L, Killander D, Lewis SE, Andersson A, et al. TRPA1 mediates spinal antinociception induced by acetaminophen and the cannabinoid Delta(9)-tetrahydrocannabiorcol. Nat Commun. 2011;2:551.
- Dodson T. Paracetamol is an effective drug to use for pain following oral surgery. Evid Based Dent. 2007;8(3):79-80.
- Vargas-Schaffer G. Is the WHO analgesic ladder still valid? Twenty-four years of experience. Can Fam Physician. 2010 Jun;56(6):514-7, e202-5.
36. Hersh EV, Kane WT, O’Neil MG, Kenna GA, Katz NP, Golubic S, Moore PA. Prescribing recommendations for the treatment of acute pain in dentistry.
Compend Contin Educ Dent. 2011 Apr;32(3):22, 24-30
37. Minsk L Diagnosis and treatment of acute periodontal conditions. Compend Contin Educ Dent. 2006 Jan;27(1):8-11
38. Buonavoglia A, Latronico F, Pirani C, Greco MF, Corrente M, Prati C. Symptomatic and asymptomatic apical periodontitis associated with red complex bacteria: clinical and microbiological evaluation. Odontology. 2011 Dec 6. [Epub ahead of print]
- Villalobos-Rodelo JJ, Medina-Solís CE, Maupomé G, Lamadrid-Figueroa H, Casanova-Rosado AJ, Casanova-Rosado JF, Márquez-Corona Mde L. Dental needs and socioeconomic status associated with utilization of dental services in the presence of dentalpain: a case-control study in children. J OrofacPain. 2010 Summer;24(3):279-86
40. Goes, P.S.A. and Watt, R.G. and Hardy, R. and Sheiham, A. (2007) The prevalence and severity of dental pain in 14-15 year old Brazilian schoolchildren. Community Dental Health, 24 . pp. 217-224.
40. Abbott PV. Selective and intelligent use of antibiotics in endodontics. Aust End J 2000;26:30-9.
41.Arias-Irimia O, Barona-Dorado C, Santos-Marino JA, Martínez-Rodriguez N, Martínez-González JM. Meta-analysis of the etiology of odontogenic maxillary sinusitis. Med Oral Patol Oral Cir Bucal. 2010 Jan 1;15(1):e70-3
42.Wormald PJ. Treating acute sinusitis. Aust Prescr 2000;23:39-42
43.Campbell CM, Stout BM, Deas DE Necrotizing ulcerative gingivitis: a discussion of four dissimilar presentations. Tex Dent J. 2011 Oct;128(10):1041-51

